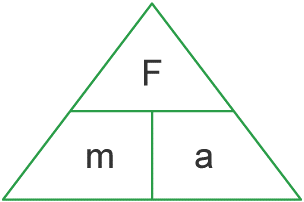
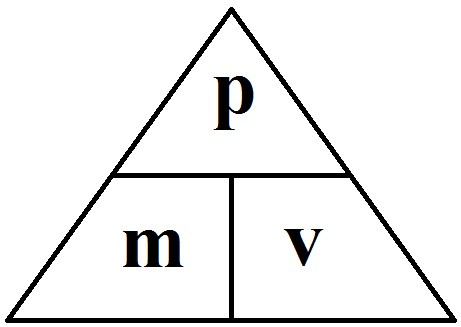
Atomic number: The number of protons in an atom.
Mass number: The number of protons and neutrons in an atom.
Isotopes: Atoms of the same element (same number of protons) but with a different number of neutrons.
Relative atomic mass (Ar): The average mass of an atom compared to 1/12th the mass of carbon-12.