Calculations practice – Molecular formulae
Refresh the page to get a different random selection of questions and answers.
Calculations practice – Reacting masses
Refresh the page to get a different random selection of questions and answers.
Calculations practice – Mixed calculations
Refresh the page to get a different random selection of questions and answers.
Calculate reacting masses from experimental data – QUIZ
[WpProQuiz 63]
[WpProQuiz 62]
[WpProQuiz 64]
[WpProQuiz 61]
[WpProQuiz 65]
[WpProQuiz 60]
Tricks for remembering the names of organic molecules
A good way to remember the names of organic molecules is to make up a silly mnemonic where the first letter of each word matches the first letter of the organic molecules. For example the first 10 alkanes in order are , Methane, Ethane, Propane, Butane, Pentane, Hexane, Heptane, Octane, Nonane and Decane. These can be memorised with “Many elephants prefer blue pinapples. However hungry orangutans never do.” This isn’t a very good mnemonic, but it is the one I made up for myself when I was at school, and you are best making up your own. The sillier, smellier and more colourful the better.
Diffusion – video
This video is a great way to see the diffusion of particles in action:
Harry Potter does The Elements – video
Watch Daniel Radcliffe sing the names of all the elements – it’s just a shame there are now more elements than were written into Tom Lehrer’s famous song…..
How do we separate the seemingly inseperable? – video
There are several things in this video which are “beyond the exam spec” but there are loads of interesting bits…..
Preparation of CuSO₄ – video
Below is the preparation of copper sulfate crystals (CuSO4.5H2O) through the process of filtration and crystallisation
1:14a know what is meant by the terms atom
Atom: An atom is the smallest part of an element.
Atomic structure – Tyler de Witt video
This video explains the basics of atomic structure, telling you what is inside an atom:
Build an atom – interactive
This is a good interactive demonstration showing how subatomic particles make up the atoms in the Periodic Table.
1:16a know what is meant by the terms atomic number, mass number and relative atomic mass (Aᵣ)
Atomic number: The number of protons in an atom.
Mass number: The number of protons and neutrons in an atom.
Relative atomic mass (Ar): The average mass of an atom compared to 1/12th the mass of carbon-12.
Calculation of relative atomic mass – Tyler de Witt video
This excellent video from Tyler de Witt walks you through what isotopes are, and how the relative abundance of those isotopes can be used to calculate the relative atomic mass of an element.
1:20a understand how to use electrical conductivity to classify elements as metals or non-metals
Metals
- conduct electricity
Non – Metals
- do not conduct electricity (except for graphite)
Balancing equations – Tyler de Witt videos
This excellent Tyler de Witt video is an introduction to balancing equations:
And here’s another of the lovely Tyler’s videos with some practice questions and answers on equation balancing:
Balancing equations – interactive
This is useful to help you to practice how to balance equations:
Calculation of relative formula mass – Tyler de Witt video
Here’s an excellent Tyler de Witt video explaining how to calculate the relative formula mass of compounds with:
- a simple formula
- a formula which includes brackets
- a formula which includes a dot (water of crystallisation)
Calculations involving mass (in grams), amount (in moles) and relative atomic mass – Tyler de Witt video
This video shows how to perform calculations involving mass (in grams), amount (in moles) and relative atomic mass:
Calculate Reacting Masses video
This video steps through a very useful method used to calculate reacting masses.
1:31a understand how the formulae of simple compounds can be obtained experimentally, including metal oxides
Finding the formula of a metal oxide experimentally
The formulae of metal oxides can be found experimentally by reacting a metal with oxygen and recording the mass changes.
Example: When magnesium is burned in air, it reacts with oxygen (O2) to form magnesium oxide (MgO).
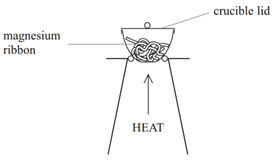
Method:
• Weigh a crucible and lid
• Place the magnesium ribbon in the crucible, replace the lid, and reweigh
• Calculate the mass of magnesium
(mass of crucible + lid + Magnesium – mass of crucible + lid)
• Heat the crucible with lid on until the magnesium burns
(lid prevents magnesium oxide escaping therefore ensuring accurate results)
• Lift the lid from time to time (this allows air to enter)
• Stop heating when there is no sign of further reaction
(this ensures all Mg has reacted)
• Allow to cool and reweigh
• Repeat the heating , cooling and reweigh until two consecutive masses are the same
(this ensures all Mg has reacted and therefore the results will be accurate)
• Calculate the mass of magnesium oxide formed (mass of crucible + lid + Magnesium oxide – mass of crucible + lid)
Empirical formulae and Molecular formulae – Tyler de Witt video
In this video, the lovely Tyler de Witt explains Empirical Formulae and Molecular formulae.
(Please excuse the horrid error right at the start where the video shows a diagram of prop-2-ene, but it is labelled ‘ethene’ by mistake.)
This second video then explains how, if given an empirical formula and a molecular mass, you can calculate the molecular formula:
Concentration (molarity) practice problems – Tyler de Witt video
Here is a video to help you with concentration calculations. In Chemistry, concentration is know as molarity (i.e. the amount per volume).
Note that in the video, the lovely Tyler de Witt uses the unit “liter” for volume, but the correct international unit (as used by UK exam boards) is decimeters cubed (dm3) which is the same thing as Tyler’s “liter”.
and a follow up video with some more examples:
What is an ion? – Tyler de Witt video
Knowing the answer to “What’s an ion?” is absolutely key to Chemistry.
If you aren’t sure what an ion is, or why the idea of ions is crucial to Chemistry, then you should watch this video:
Dogs teach ionic versus covalent bonding – video
This should give you a ruff idea of what the difference is between ionic and covalent bonding.
In an ion, the number of protons does not equal the number of electrons – Tyler de Witt video
In an electrically neutral ATOM, the number of electrons equals the number of protons.
However, an ION is an atom (or group of atoms) which has either gained or lost some electrons, so for an ION the number of electrons does not equal the number of protons.
This excellent Tyler de Witt video explains this clearly:
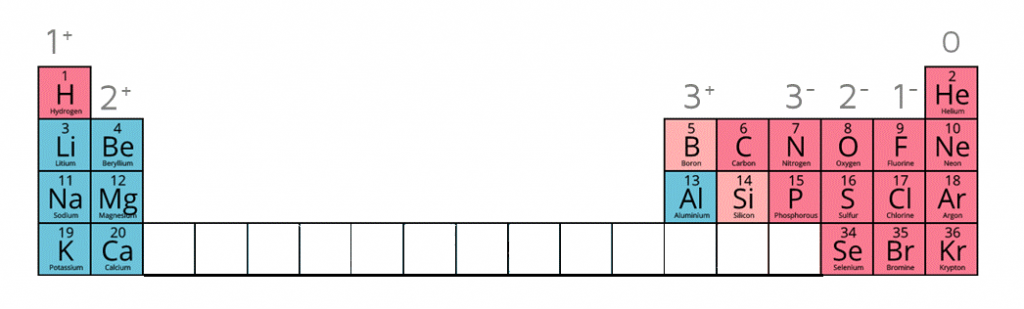
1:38a understand how to use the charges of these ions in ionic formulae: metals in Groups 1, 2 and 3, non-metals in Groups 5, 6 and 7, Ag⁺, Cu²⁺, Fe²⁺, Fe³⁺, Pb²⁺, Zn²⁺, hydrogen (H⁺), hydroxide (OH⁻), ammonium (NH₄⁺), carbonate (CO₃²⁻), nitrate (NO₃⁻), sulfate (SO₄²⁻)
When given this information of the following ions, it is possible to work out the formulae of ionic compounds which include these ions.
| Name of Ion | Formula | Charge |
|---|---|---|
| Sulfate | SO42- | -2 |
| Carbonate | CO32- | -2 |
| Nitrate | NO3- | -1 |
| Hydroxide | OH- | -1 |
| Ammonium | NH4+ | +1 |
| Silver ion | Ag+ | +1 |
| Zinc ion | Zn2+ | +2 |
| Hydrogen ion | H+ | +1 |
| Copper (II) ion | Cu2+ | +2 |
| Iron (II) ion | Fe2+ | +2 |
| Iron (III) ion | Fe3+ | +3 |
| Lead (II) ion | Pb2+ | +2 |
Ion charges on the periodic table