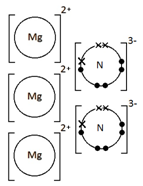
1:40 draw dot-and-cross diagrams to show the formation of ionic compounds by electron transfer, limited to combinations of elements from Groups 1, 2, 3 and 5, 6, 7 only outer electrons need be shown
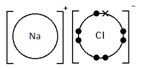
Sodium chloride, NaCl
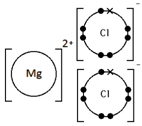
Magnesium chloride, MgCl2
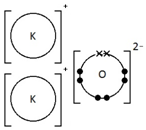
Potassium oxide, K2O
Calcium oxide, CaO
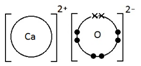
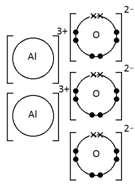
Aluminium oxide, Al2O3
Magnesium nitride, Mg3N2