3:19 (Triple only) know that a reversible reaction can reach dynamic equilibrium in a sealed container
A dynamic equilibrium can only be achieved in a sealed container so substances cannot escape.
A dynamic equilibrium can only be achieved in a sealed container so substances cannot escape.
Features of a reaction mixture that is in dynamic equilibrium:
A catalyst is a substance which increases the rate of reaction without being chemically changed at the end of the reaction.
A reversible reaction is one where the forward reaction and the backward reaction happen simultaneously. For example:
3H₂ + N₂ ⇋ 2NH₃
In such a reaction a catalyst speeds up both the forward and the backward reactions. Hence, although the system will reach dynamic equilibrium more quickly, the addition of a catalyst will not affect the position of equilibrium.
In a reversible reaction the position of the equilibrium (the relative amounts of reactants and products) is dependent on the temperature and pressure of the reactants.
If the conditions of an equilibrium reaction are changed, the reaction moves to counteract that change.
Therefore by altering the temperature or pressure the position of the equilibrium will change to give more or less products.
Adding a catalyst does not affect the position of the equilibrium.
If a change in conditions moves equilibrium to the right, the yield of the substances on the right is increased.
Changing the temperature:
All reactions are exothermic in one direction and endothermic in the other way.
For this reaction the enthalpy change, ΔH is negative therefore the forward reaction is exothermic:
CO(g) + 2H2(g) ⇌ CH3OH(g) ΔH = –91 kJ/mol
If temperature is decreased the position of the equilibrium will shift to the right because it is an exothermic reaction.
For this reaction the enthalpy change, ΔH is positive therefore the forward reaction is endothermic:
CH4(g) + H2O(g) ⇌ CO(g) + 3H2(g) ΔH = +210 kJ mol–1
If temperature is increased the position of the equilibrium will shift to the right because it is an endothermic reaction.
Key point: an increase (or decrease) in temperature shifts the position of equilibrium in the direction of the endothermic (or exothermic) reaction
Changing the pressure:
Reactions may have more molecules of gas on one side than on the other.
For this reaction there are 2 molecules on the left and 4 molecules on the right:
CH4(g) + H2O(g) ⇌ CO(g) + 3H2(g) ΔH = +210 kJ mol–1
If the pressure is increased the position of the equilibrium will shift to the left because there are fewer molecules on the left-hand side.
For this reaction there are 3 molecules on the left and 1 molecule on the right
CO(g) + 2H2(g) ⇌ CH3OH(g) ΔH = –91 kJ/mol
If the pressure is decreased the position of the equilibrium will shift to the left because there are more molecules on the left-hand side.
Key point: an increase (or decrease) in pressure shifts the position of equilibrium in the direction that produces fewer (or more) moles of gas
This video explains how the position of equilibrium in a reversible reaction is affected by changes to temperature.
This video explains how the position of equilibrium in a reversible reaction is affected by changes to pressure.
Hydrocarbon: A molecule containing only hydrogen and carbon
The molecular formula shows the actual number of atoms of each element in a molecule.
The general formula shows the relationship between the number of atoms of one element to another within a molecule. Members of a homologous series share the same general formula. The general formula for alkanes is CnH2n+2 and the general formula for alkenes is CnH2n.
A structural formula shows how the atoms in a molecule are joined together.
The displayed formula is a full structural formula which shows all the bonds in a molecule as individual lines.
The terms above are demonstrated with the example of butane.
The terms above are demonstrated with the example of ethene, which contains a double bond.
The molecular formula shows the actual number of atoms of each element in a molecule.
The empirical formula shows the simplest whole number ratio of atoms present in a compound. So the molecular formula is a multiple of the empirical formula.
The general formula shows the relationship between the number of atoms of one element to another within a molecule. Members of a homologous series share the same general formula. The general formula for alkanes is CnH2n+2 and the general formula for alkenes is CnH2n.
A structural formula shows how the atoms in a molecule are joined together.
The displayed formula is a full structural formula which shows all the bonds in a molecule as individual lines.
The terms above are demonstrated with the example of butane.
The terms above are demonstrated with the example of ethene, which contains a double bond.
Isomers are molecules with the same molecular formula but with a different structure.
A functional group is an atom or a group of atoms that determine the chemical properties of a compound.
For example the functional group of an alcohol is the -OH group and that of alkenes is the C=C carbon to carbon double bond.
A Homologous series is a group of substances with:
Isomers are molecules with the same molecular formula but with a different structure.
The names of organic molecules are based on the number of carbon atoms in the longest chain. This chain is the longest consecutive line of carbon atoms, even if this line bends.
| The name is based on the number of carbon atoms in the longest chain |
|---|
| 1 Meth- |
| 2 Eth- |
| 3 Prop- |
| 4 But- |
| 5 Pent- |
| 6 Hex- |
| 7 Hept- |
| 8 Oct- |
| 9 Non- |
| 10 Dec- |
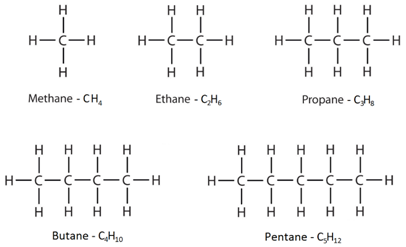
Hydrocarbons are molecules which contain only hydrogen and carbon.
Naming straight-chain alkanes
The simplest hydrocarbons are alkanes. They contain only single bonds, and have “-ane” in the name.
For example, the displayed formula of ethane (C₂H₆) is:
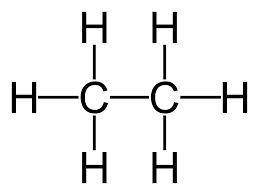
The name “ethane” contains “eth-” because there are 2 carbon atoms in the longest chain, and the name contains “-ane” because the molecule only has single bonds so is an alkane.
Another example is pentane (C₅H₁₂) which has the displayed formula:
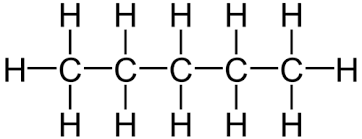
The name “pentane” contains “pent-” because there are 5 carbon atoms in the longest chain, and the name contains “-ane” because the molecule only has single bonds, so is an alkane.
Remember, it does not matter if the longest consecutive line of carbons bends around. For example the displayed formula below still shows a very normal molecule of pentane (5 carbons in a row). Pentane is not normally drawn with the longest chain of carbons bent around because it could be confusing.
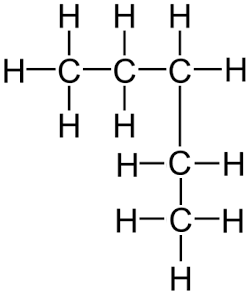
You might also see the bonds drawn at angles. Don’t worry, the displayed formula below is still pentane, as can be seen by the fact there are 5 carbon atoms in the longest chain, surrounded by hydrogen atoms bonded to the carbon atoms by single bonds.
A shorter way to express the detailed structure of an organic molecule is the structural formula. The structural formula for pentane is CH₃-CH₂-CH₂-CH₂-CH₃, which tells us the same information about the molecule as does the displayed formula, without the hassle of having to draw all the bonds or all the hydrogen atoms.
Naming straight-chain alkenes
Another simple group of hydrocarbons is the alkenes. They contain a carbon-to-carbon double bond, which also means they have two fewer hydrogen atoms than their corresponding alkane. An alkene has “-ene” in its name.
For example, the displayed formula for ethene (C₂H₄) is:
and the displayed formula of propene (C₃H₆) is:
With longer alkene molecules the double bond might appear in different locations of the carbon chain, so the name needs to be a little bit more complicated to be able to describe these differences clearly. A number is added in the middle of the name to indicate at which carbon the double bond starts.
So the displayed formula of pent-1-ene is:
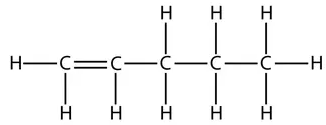
and this is the displayed formula of pent-2-ene:
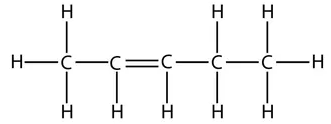
However, take care that when counting which carbon has the double bond. The numbers start from the end that produces the smallest numbers in the name. For example, this is the displayed formula for pent-1-ene again, but just drawn the other way round. It is still pent-1-ene (you can’t get pent-4-ene):
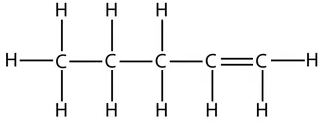
Naming straight-chain alcohols
We get the same pattern all over again with the group of organic molecules called alcohols, which are recognised by an -OH functional group. For example here is the displayed formula for ethanol, which has 2 carbon atoms in the longest chain:
and here is the displayed formula for butanol:
Summary of naming simple straight-chain organic molecules
The following table summarises the naming of some of the straight-chain alkanes, alkenes and alcohols, giving a name and a molecular formula for each:
| Carbons in longest chain | Alkanes | Alkenes | Alcohols |
|---|---|---|---|
| 1 | methane, CH₄ | - | methanol, CH₄O |
| 2 | ethane, C₂H₆ | ethene, C₂H₄ | ethanol, C₂H₆O |
| 3 | propane, C₃H₈ | propene, C₃H₆ | propanol, C₃H₈O |
| 4 | butane, C₄H₁₀ | butene, C₄H₈ | butanol, C₄H₁₀O |
Naming branched alkanes and alkenes
The naming conventions for organic molecules cover more than the straight chain molecules. Branched molecules are named depending on the number of carbon atoms in the branch. A branch with 1 carbon is called “methyl” and a branch with 2 carbons is called “ethyl”. This is similar to the conventions covered above, plus the “-yl-” bit just says it is a branch.
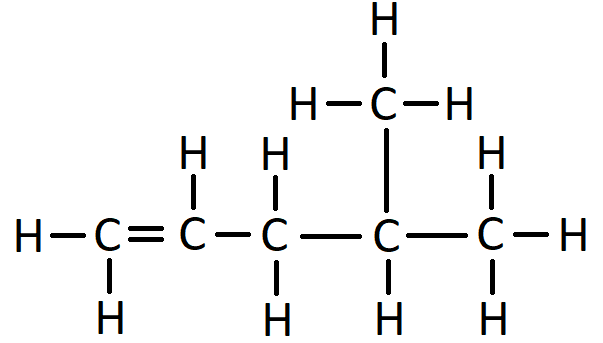
For example, this is the displayed formula for 2-methyl hexane:
In the name 2-methyl hexane, the number 2 indicates that when counting along the longest carbon chain the methyl branch comes off the second carbon atom. The “methyl” bit of the name says there is one branch of 1 carbon. The “hex” bit of the name says the longest consecutive chain of carbon atoms is 6. The “ane” bit says the molecule has only single bonds.
When counting along the carbon atoms of the longest chain to work out the name, the numbering of carbon atoms starts from the end nearest to the branch. Another way to put this is that the name is given such that the numbers in the name are as low as possible. For example, here is the displayed formula for 4-ethyl octane:
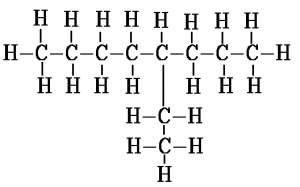
Another example, this time with 2 methyl branches coming off the second and third carbons of the chain, is 2,3-dimethyl hexane. The “di” in the name indicates there are two methyl groups. This is the same way in which “di” indicates there are two oxygen atoms in carbon dioxide.
Another example of how the naming convention works for branches is 2,2-dimethyl hexane:
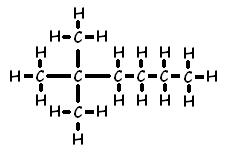
This naming of branches also applies to alkenes. Here is the displayed formula of 4-methylpent-1-ene:
The molecular formula describes the actual number of each type of each atom in a molecule.
For example, a molecular formula of C₆H₁₂ tells us that in each molecule there are 6 carbon atoms and 12 hydrogen atoms.
However, the molecular formula tells us nothing about how those atoms are arranged. For example, it does not tell us if there any branches of carbon atoms coming off the main carbon-carbon chain, nor how long or how many there might be.
On the other hand, the structural formula and displayed formula of a molecule tell us clearly how the atoms are arranged in that molecule.
This means that if we are given a molecular formula only, there may be several possible structural and displayed formulae all of which could apply for that molecule.
When trying to work out possible structural or displayed formulae from a molecular formula there are several clues:
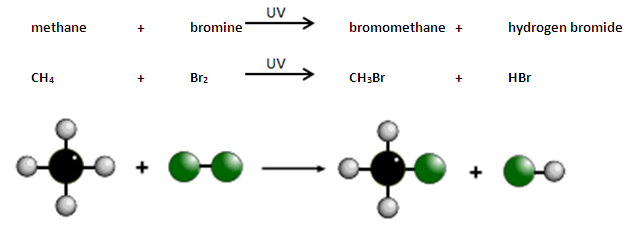
In a substitution reaction an atom or group of atoms is replaced by a different atom or group of atoms. For example when ethane reacts with bromine gas one of the hydrogen atoms in ethane is substituted by one of the atoms of bromine from within the bromine molecule:
CH₃-CH₃ + Br-Br → CH₃-CH₂Br + H-Br
ethane + bromine → bromoethane + hydrogen bromide
An addition reaction occurs when an atom or group of atoms is added to a molecule without taking anything away. For example when ethene reacts with bromine gas, the product is simply the addition of the two molecules:
CH₂=CH₂ + Br-Br → CH₂Br-CH₂Br
A combustion reaction is another way to say ‘burning’ and is a reaction with oxygen. Combustion of hydrocarbons with excess oxygen gives the products water and carbon dioxide, and also releases heat energy (exothermic reaction). Two examples the combustion of propane and the combustion of butene:
C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
C₄H₈ + 6O₂ → 4CO₂ + 4H₂O
This video shows the addition reaction between bromine and an alkene.
The observation from the reaction is the colour change from orange to colourless.
Crude Oil is a mixture of hydrocarbons.
This video does not quite use the right language for the various fractions as appropriate to the Edexcel iGCSE, but it is nevertheless a good description of the process. Make sure you use the notes on tutorMyself.com to get the exact language you will need for your exam.
And another somewhat older video showing the industrial process of fractional distillation:
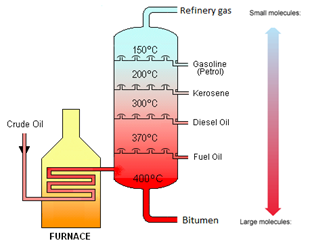
Crude oil is separated into fractions by the process of fractional distillation.
| Fraction | Use |
|---|---|
| Refinery gases | Bottled gas |
| Gasoline | Fuel for cars |
| Kerosene | Fuel for aeroplanes |
| Diesel Oil | Fuel for lorries |
| Fuel Oil | Fuel for ships |
| Bitumen | Road Surfacing |
The boiling point increases as the number of carbon atoms (chain length) increases.
The viscosity increases as the number of carbon atoms (chain length) increases.
The greater the number of carbon atoms (chain length), the darker in colour that fraction is.
The viscosity of a fluid describes how easily it flows. Water has a low viscosity, it flows very easily. Crude oil has a higher viscosity than water, it does not flow very easily.
| Fractions (in order) | Properties |
|---|---|
| Refinery gases | Smallest molecules. Lowest boiling point. Lowest viscosity. Lightest in colour. |
| Gasoline | |
| Kerosene | |
| Diesel | |
| Fuel oil | |
| Bitumen | Largest molecules. Highest boiling point. Highest viscosity. Darkest in colour. |
A fuel is a substance that, when burned, releases heat energy (exothermic reaction).
Complete Combustion happens when there is enough oxygen available, producing carbon dioxide (CO2) and water (H2O)
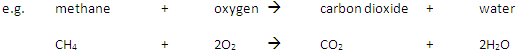
Incomplete Combustion happens when there is not enough oxygen available, with possible products being carbon monoxide (CO), carbon (C, soot), carbon dioxide (CO2) and water (H2O)
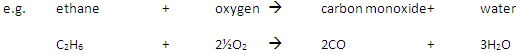
Carbon monoxide may be produced from the incomplete combustion of fuels:
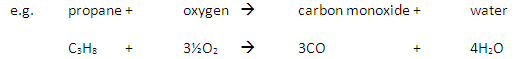
Carbon monoxide is poisonous because it reduces the capacity of the blood to carry oxygen.
When fuels are burned in vehicle engines, high temperatures are reached.
At these high temperatures nitrogen and oxygen from the air react to produce nitrogen oxides:
nitrogen + oxygen → nitrogen oxides
eg
N2 (g) + O2 (g) → 2NO (g)
In the atmosphere these nitrogen oxides can combine with water to produce nitric acid (HNO3).
Fossil fuels such as coal, gas and oil are derived from crude oil.
These fuels are hydrocarbons, but also include impurities such as sulfur.
When the fuels are burned, sulfur dioxide is produced which can escape into the atmosphere:
S (s) + O₂ (g) → SO₂ (g)
Acids formed in the atmosphere can fall as acid rain. This can be a major problem, killing trees and fish in lakes. The acid rain also corrodes limestone buildings and marble statues since these are both made of calcium carbonate (CaCO₃). Some metals such as iron are also attacked by acid rain.
Sulfur dioxide released into the atmosphere from the burning of fossil fuels can react with water and oxygen to make sulfuric acid (H₂SO₄):
2SO₂ (g) + 2H₂O (l) + O₂ (g) → 2H₂SO₄ (aq)
Also, if sulfur dioxide in the atmosphere reacts with just water, a weaker acid called sulfurous acid (H₂SO₃) is formed:
SO₂ (g) + H₂O (l) → H₂SO₃ (aq)
In car engines the temperature is high enough for the nitrogen in the air to react with oxygen to produce oxides of nitrogen, e.g:
N₂ (g) + O₂ (g) → NO₂ (g)
In the atmosphere these nitrogen oxides can produce nitric acid (HNO₃).
Cracking involves the thermal decomposition of long-chain alkanes into shorter-chain alkanes and alkenes:
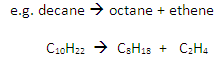
Conditions
Temperature: 600oC
Catalyst: aluminium oxide, Al2O3
this somewhat old video talks about the industrial process of cracking:
Cracking converts long chain hydrocarbons into short chain hydrocarbons.
Long-chain alkanes are broken down into alkanes and alkenes of shorter length.
Crude oil contains a surplus long chains.
Shorter chain hydrocarbons are in greater demand, e.g. petrol.
Cracking also produces alkenes which are used in making polymers and ethanol.
Alkanes have the general formula CnH2n+2
This means that to work out the number of hydrogens, you double the number of carbons and then add two.
Saturated: A molecule containing only single bonds between carbon atoms. For example, alkanes as described as saturated molecules.
Unsaturated: A molecule containing a carbon-carbon double or triple bond. For example, alkenes as described as unsaturated molecules.
The displayed formulae show all the atoms and bonds drawn out.
The molecular formulae just show the number of each type of atom in the molecule.
This video is a good introduction to alkanes and crude oil:
Alkanes react with bromine in the presence of UV light, e.g. sunlight.
A hydrogen atom in the alkane is replaced by a bromine atom.
This is known as substitution.
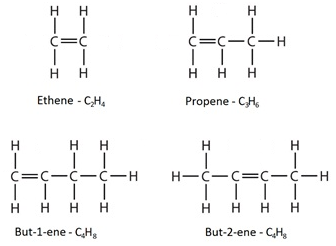
Alkenes are a homologous series of hydrocarbons which contain a carbon-carbon double bond. This double bond is shown in formulae as a double line.
The names of alkenes end with “ene”.
An example is ethene, the structural formula for which is CH₂ = CH₂
For a molecule with more than two carbon atoms, the position of the double bond within the molecule can vary as indicated by the name and the structural formula.
Alkenes have the general formula CnH2n
So an alkene always has twice as many hydrogen atoms as carbon atoms.
Saturated: A molecule containing only single bonds between carbon atoms. For example, alkanes as described as saturated molecules.
Unsaturated: A molecule containing a carbon-carbon double or triple bond. For example, alkenes as described as unsaturated molecules.
The displayed formulae show all the atoms and bonds drawn out.
The molecular formulae just show the number of each type of atom in the molecule.
Alkenes react with bromine water. UV light is not required for this reaction.
The double bond is broken and the bromine atoms are added. This is an addition reaction.
During this reaction there is a colour change from orange to colourless.
For example:
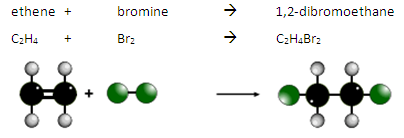
This is how we can test for the presence of an alkene or another type of unsaturated molecule.
In the absence of UV light an alkane added to bromine water will not react: the bromine water will stay orange.
However, alkenes react with bromine water even without UV light. There will be a colour change of orange to colourless.
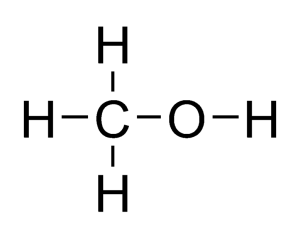
The member of the homologous series called Alcohols have names which end in “ol”. Examples are methanol, ethanol and propanol.
Alcohols all contain an -OH functional group attached to a hydrocarbon chain.
Structural formula and displayed formula for methanol:
CH₃-OH
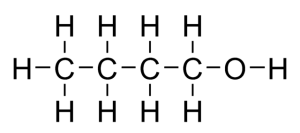
Structural formula and displayed formula for ethanol:
CH₃-CH₂-OH (or simply C₂H₅OH)
Structural formula and displayed formula for propan-1-ol:
CH₃-CH₂-CH₂-OH
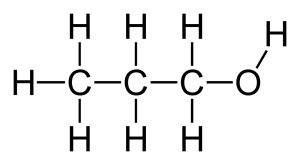
Structural formula and displayed formula for butan-1-ol:
CH₃-CH₂-CH₂-CH₂-OH
This video introduces alcohols:
1) Ethanol can be oxidised by complete combustion. With excess oxygen the complete combustion of ethanol (C₂H₅OH) in air produces carbon dioxide and water:
C₂H₅OH (l) + 3O₂ (g) → 2CO₂ (g) + 3H₂O (l)
2) Ethanol can be oxidised in air in the presence of microorganisms (‘microbial oxidation’) to form ethanoic acid (CH₃COOH).
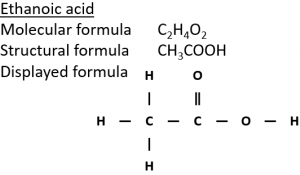
3) Ethanol can be oxidised by heating with the oxidising agent potassium dichromate(VI) (K₂Cr₂O₇) in dilute sulfuric acid (H₂SO₄).
In the equation below, [O] means oxygen from an oxidising agent.
CH₃CH₂OH + 2[O] → CH₃COOH + H₂O
This mixture starts orange but when the reaction happens turns green which indicates the presence of Cr³⁺ ions which are formed when the potassium dichromate(VI) is reduced.
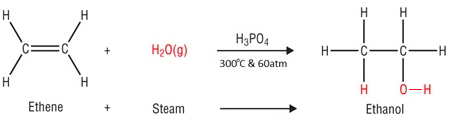 In the hydration of ethene, ethanol is made by passing ethene and steam over a catalyst.
In the hydration of ethene, ethanol is made by passing ethene and steam over a catalyst.
Water is added to ethene, this is known as hydration.
Conditions
Catalyst: Phosphoric acid (H3PO4)
Temperature: 300°C
Pressure: 60 atm
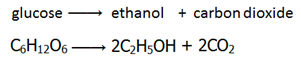 Fermentation is the conversion of sugar, e.g. glucose into ethanol by enzymes from yeast.
Fermentation is the conversion of sugar, e.g. glucose into ethanol by enzymes from yeast.
Conditions
Catalyst: Zymase (enzyme found in yeast)
Temperature: 30°C – The process is carried out at low temperatures as not to denature the enzymes.
Other: Anaerobic (no oxygen present) – if oxygen were present, the yeast produce carbon dioxide and water instead of ethanol.
In the production of ethanol the process of fermentation is carried out at a low temperature (30⁰-40⁰).
Above 40⁰ the enzymes would permanently lose their structure (denature).
At a temperature lower than 30⁰ the process would be too slow.
Fermentation is conducted in the absence of air. In the presence of air (aerobic conditions), enzymes in the yeast produce carbon dioxide and water instead of ethanol.
Also, in the presence of air, the ethanol can oxidise to ethanoic acid.